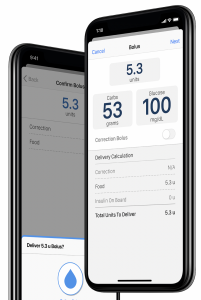
Tandem Diabetes Care, Inc., a technology company, leader in insulin delivery and diabetes, announced U.S. Food and Drug Administration (FDA) clearance of bolus insulin dosing on the t:slim X2 insulin pump using the t:connect mobile app.
This represents the first-ever FDA-cleared smartphone app capable of initiating insulin delivery. The mobile app can work on both iOS and Android operating systems. The updated t:connect app is designed to offer t:slim X2 insulin pump users the ability to program and cancel bolus insulin requests. John Sheridan, president and CEO of Tandem Diabetes Care, said:
“This FDA clearance further validates our commitment to innovation and the diabetes community by providing one of the most requested feature enhancements. With the improvements in diabetes management provided by Tandem’s Control-IQ technology, giving a meal bolus is now the most common reason a person interacts with their pump, and the ability to do so using a smartphone app offers a convenient and discrete solution.”
The new feature will be offered in the United States for free to new t:slim X2 insulin pump customers and to in-warranty customers through a remote software update for the t:slim X2 insulin pump and the updated t:connect mobile app.
The Company plans to roll out the mobile feature update throughout the spring in a series of limited launch groups, followed by an expanded launch later during the summer. Limited launch participants have already been selected.
The t:connect mobile app is a mobile application that, when paired with a t:slim X2 insulin pump, discreetly displays the user’s pump information on their phone. What is more, the mobile app displays the last 24 hours of glucose trends, pump status changes, and insulin therapy data, including basal and bolus deliveries and suspensions of insulin delivery.
The app also shows pump alerts and alarms, and securely uploads data into the cloud-based t:connect web application.
It is important to know that the t:slim X2 insulin pump works independently from the t:connect mobile app, hence, a user retains the freedom to view pump therapy data, program requests and cancel bolus insulin requests from their pump.
The t:connect mobile app is compatible with select versions of both iOS and Android operating systems in the United States. For better compatibility, Tandem verifies each combination of device and operating system works as intended to program and cancel a bolus from your smartphone.
Moreover, Tandem was also the first to receive an interoperable automated glycemic controller designation with Control-IQ technology in December 2019, the first to receive an alternate controller-enabled infusion pump classification in February 2019, and the first insulin pump designated as compatible with integrated continuous glucose monitoring devices in June 2018.